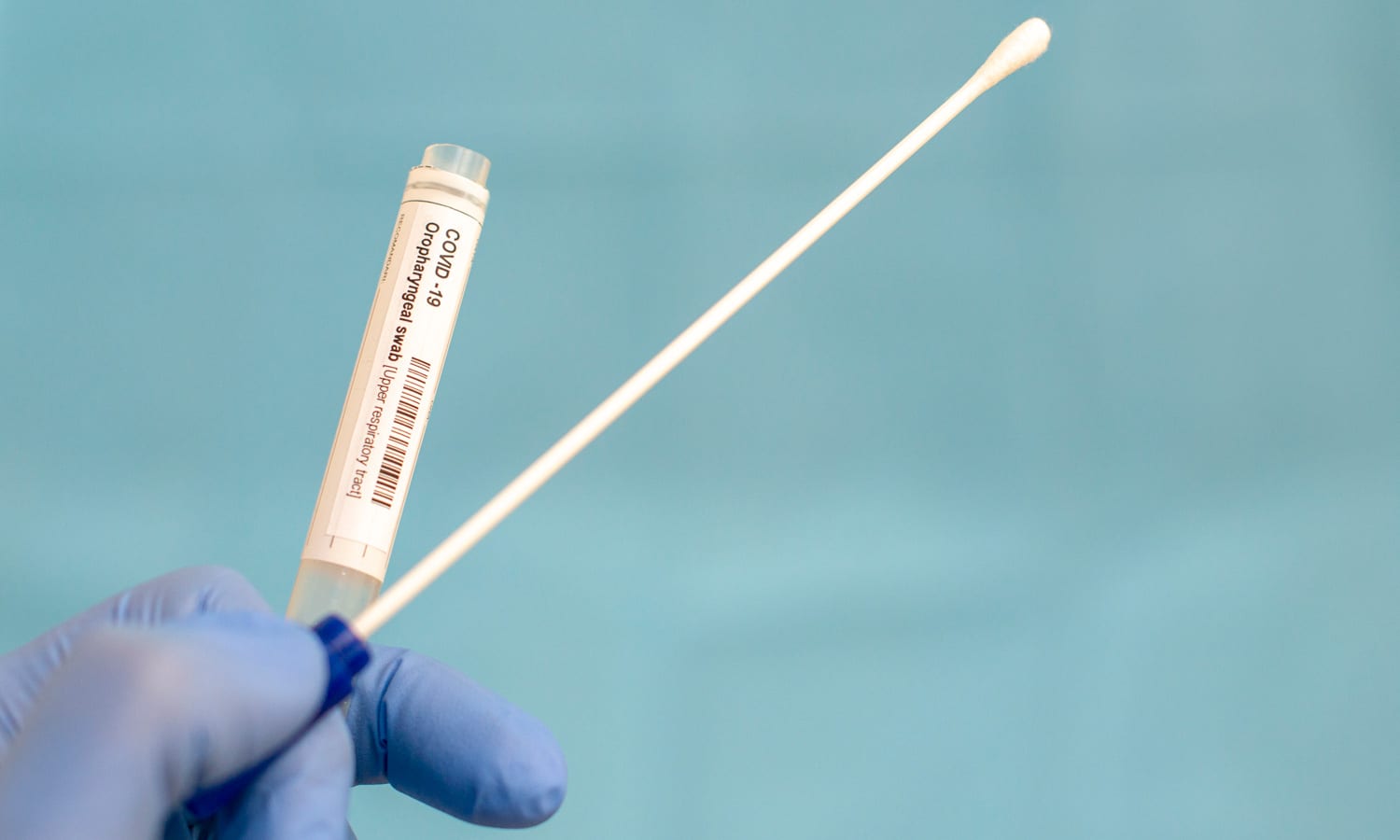
This new method of collecting saliva eliminates the risk to those handling the samples and keeps front-line laboratory workers safe in the midst of a pandemic.
“Authorizing additional diagnostic tests with the option of at-home sample collection will continue to increase patient access to testing for COVID-19. This provides an additional option for the easy, safe and convenient collection of samples required for testing without traveling to a doctor’s office, hospital or testing site.” — FDA Commissioner Stephen M. Hahn, M.D
The potential to expand the availability and accessibility of an at-home saliva test to combat COVID-19 has taken a step forward. The test approval, in a boon to the process of serology (or antibody) testing, was announced by the U.S. Food and Drug Administration (FDA) in conjunction with Accurate Diagnostic Labs (ADL), the largest independent New York-New Jersey area testing laboratory.
The FDA announcement on May 8 was an extension to the emergency use authorization (EUA), expanding it to include the saliva test. The EUA bypasses the normal labyrinthine process of government approval and authorization. This strategically important approval expands beyond the already approved at-home tests using a nasal swab and saline.
“For the past month, we have focused on providing solutions to health systems, first responders, and essential employees,” said Rupen Patel, CEO of New Jersey-based ADL. “Completing these studies and receiving this approval will allow us to continue that mission while expanding our capabilities to begin to test more people that may be spreading the virus without knowing it.”
The Rutgers Clinical Genomics Laboratory received an expanded approval for their COVID-19 laboratory developed test; this is the first saliva test to win FDA approval. This will allow testing of samples self-collected by patients at home using the Spectrum Solutions LLC SDNA-1000 saliva collection device.
With the rapid spread of COVID-19, there have been several weeks of high risk exposure to the virus — a risk that resulted in the loss of thousands of lives (including hundreds of medical personnel), ensuring that the urgency of this step forward.
RELATED: The FDA Is Taking A Big Step Toward Prevention And Treatment Of COVID-19
The new method of collecting specimens is the first-of-its-kind due to a unique way in storing the sample, explained Dr. Shaila Nayak, Medical Director of Accurate Diagnostic Labs. The design eliminates the risk to those handling the samples and keeps front-line laboratory workers safe in the midst of a pandemic.
As overall deaths from the coronavirus have passed the 100,000 mark, there are significant benefits to new specimen collection and serology breakthroughs; especially when the test mitigates biological hazards to laboratory workers.
RELATED: What Healthcare Workers Need To Know About Potential COVID-19 Treatment Remdesivir
The FDA authorization is limited to testing performed at the Rutgers Clinical Genomics Laboratory using their molecular LDT COVID-19 authorized test for saliva specimens collected using the Spectrum Solutions LLC SDNA-1000 Saliva Collection Device.
There has been a focused round-the-clock effort by all of the collaborating organizations and their research and medical personnel, together with the FDA to focus on not only finding new ways to combat the virus but add numbers to needed data patterns to understand its spread.